חלק ב' גמישות העורקים, מה אפשר לעשות
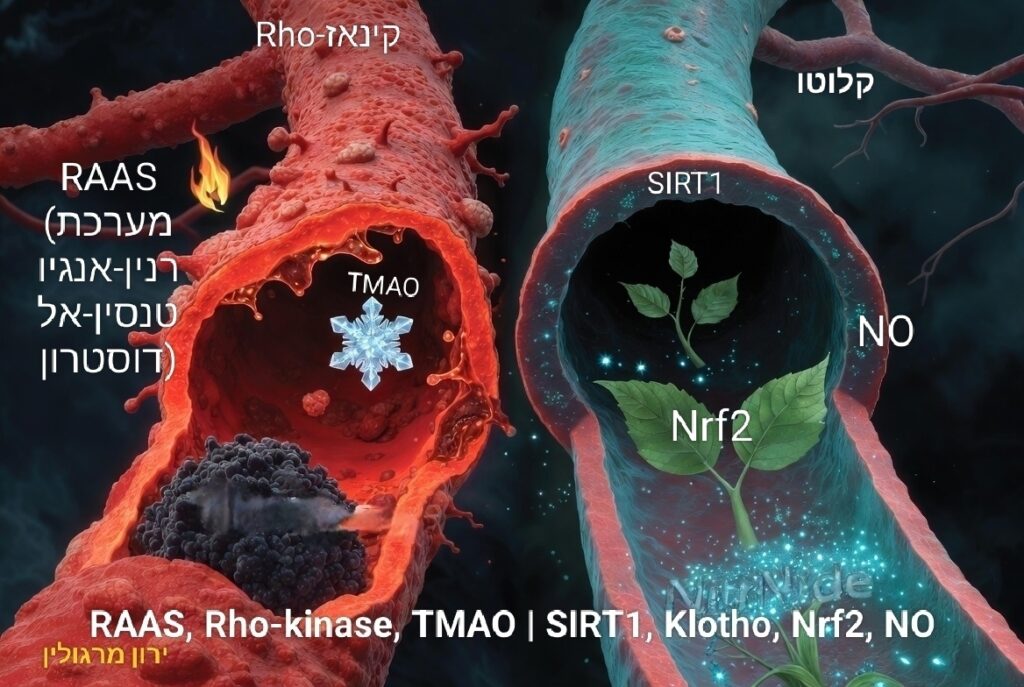
RAAS
NF‑κB, NLRP3
Nrf2
mTOR/AMPK
TMAO
ברדיקינין
ארגינין/NO (כולל הפרדוקס של המינון)
anti-GAD
VDR
RhoA/Rho‑kinase
Klotho, שמש, טלומרים
השאלה העומדת על הפרק פשוטה:
"איך מחזירים לגוף את היכולת להפעיל מחדש את מנגנוני התיקון, ההתחדשות והאיזון שלו?"
[מקור].
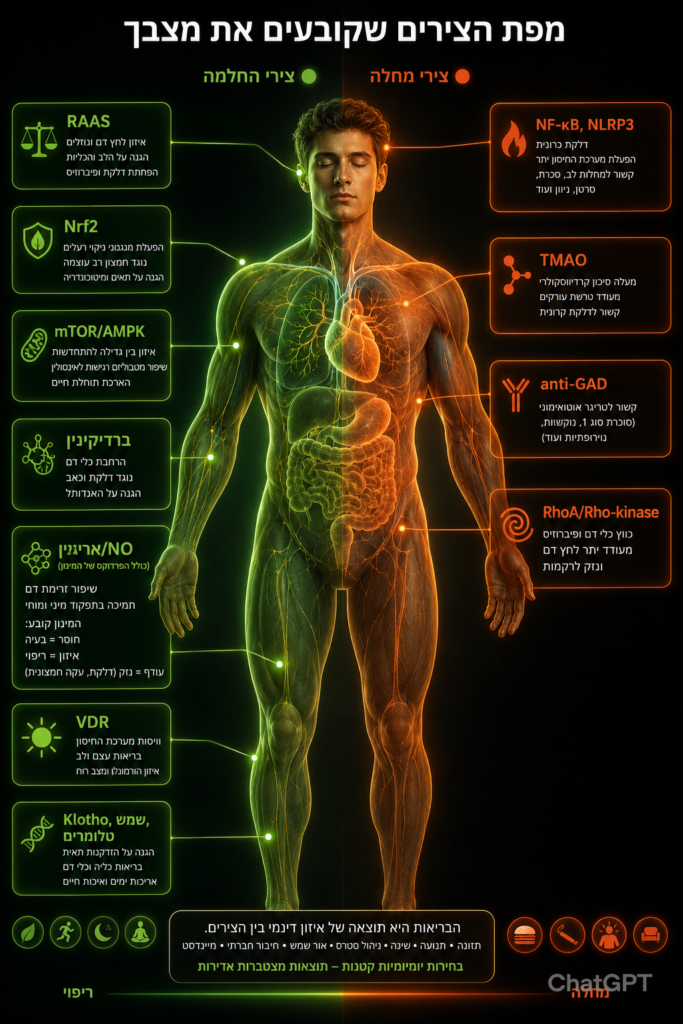
מפת הצירים שקובעים את מצבך
מה לעשות (רק באישור הרופא המטפל)
עורקים גמישים – הדרך לבריאות מחדש
העורקים הם צינורות. וכמוהם גם כל צינורות הגוף: מעברי הלימפה, צינורות הניקוז בכליות, דרכי האוויר בריאות, ומערכת השתן (כליה). כאשר השרירים בדפנות צינורות אלו מאבדים גמישות, נפגעת זרימת הנוזלים, הגוף מתקשה לפנות פסולת, ומתחיל תהליך שמקדם מחלות כרוניות: שבץ מוחי, אי-ספיקת כליות, מחלות לב, סוכרת, כבד שומני, דלקת מפרקים, ירידה קוגניטיבית, אלצהיימר ופרקינסון [מקור].
גמישות צנרת הגוף היא מדד שורש, לא רק תוצאה של מחלה. זהו מדד שיכול להציל חיים — להפחית לחץ דם, לסלק סוכרת בתוך זמן די קצר, ואפילו לקדם תהליכי שיקום מאי-ספיקת כליות ואובדן ראייה.
במקום להתמקד רק ב"ניהול מחלה" באמצעות תרופות, המאמר מציע מחשבה שנשענת על מחקרים ומקדמת גישה אחרת: שיקום צירי החלמה ביולוגיים רדומים. בהמשך המאמר יציג מפה מדעית של צירים שמקדמים נזק וצירים שמקדמים החלמה, ואת הדרכים הפרקטיות להחזיר גמישות לצינורות הגוף.
החלמה אינה תוצאה של כוח חיצוני… אלא הענקה של תנאים מדויקים שבהם הגוף חוזר לפעול [מקור].
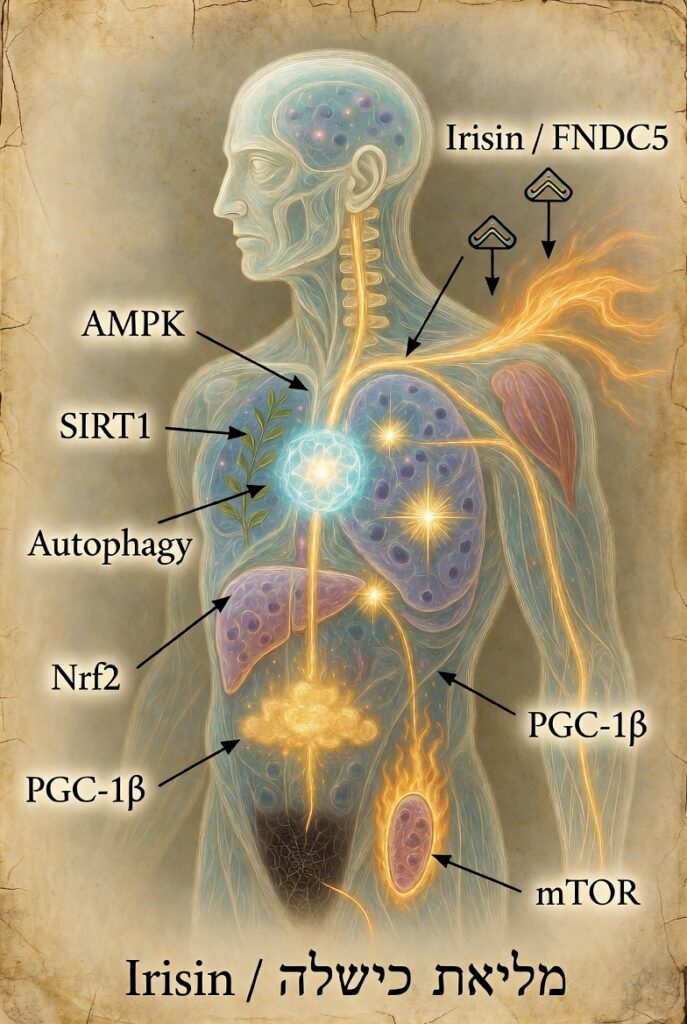
- מסלולי ההחלמה -המסלולים הביולוגיים שמשקמים את הגוף – חלק א' מאת ירון מרגולין.
- המסלולים הביולוגיים שמשקמים את הגוף – חלק ב', על ההחלמה מאת ירון מרגולין
- חלק ג' – על ההחלמה
- חלק ד' – על שומר החומר הגנטי🔹🐦🔥 P53 🐦🔥 והנחיות טבעיות של הגוף ל: "החלמה".
- חלק ה' – חלבוני ההחלמה: הרשת המולקולרית של הריפוי, ההתחדשות וההומאוסטזיס מאת ירון מרגולין
- חלק ו' על צירי ההחלמה ומה שמפעיל אותם – (signal architecture) מאת ירון מרגולין
- 🔹 מפת מסלולי ההחלמה – 3 צירים – מבט כללי
- המסלולים הביולוגיים המשקמים את🔹 הגוף: פרספקטיבה חדשה על החלמה – מבט עומק על על 3 המסלולים המרכזיים.
מפת הצירים: מסלולי נזק מול מסלולי החלמה
כדי להבין את תהליך אובדן גמישות צנרת הגוף, נוח להציג אותו כשני עולמות מתחרים:
מסלולים שמקדמים נזק, דלקת, קשיחות וסגירה של צינורות הגוף,
vs.
מסלולים שמקדמים גמישות, ניקוי, בנייה ושיקום [מקור].
להלן רשימה של הצירים העיקריים הפועלים בגוף האדם:
מפת הצירים: מסלולי נזק מול מסלולי החלמה
אתחיל בחשובים לתהליכי השיקום – אלה שצריך לחסום❌ הם שמקדמים אובדן גמישות צנרת הגוף,
מסלולים שמקדמים נזק וקשיחות
| ציר | מה הוא עושה | התוצאה |
|---|---|---|
| RAAS (רנין–אנגיוטנסין–אלדוסטרון) | מאזן נוזלים ולחץ דם; כשמופעל יתר על המידה, מעלה לחץ דם, דלקת ונזק לכלי דם | לחץ דם גבוה, טרשת, נזק לכליות |
| NF‑κB ו-NLRP3 inflammasome | מרכזיים בדלקת כרונית; מעלים ציטוקינים דלקתיים | טרשת עורקים, פגיעה בתיקון רקמות |
| eNOS uncoupling | האנזים eNOS "מתנתק" ומייצר רדיקלים חופשיים במקום NO | לחץ חמצוני, פגיעה באנדותל |
| TMAO | תוצר מטבולי מכולין וקרניטין (בשר אדום, ביצים) | טרשת עורקים, קשיחות, אירועי לב ושבץ |
| עודף ארגינין + ייצור מוגזם של NO | במינון יתר, במיוחד כשה-GFR יורד | פרוקסיניטריט, ניטרציה של חלבונים, נזק לכליות ולעורקים |
| anti-GAD | נוגדנים עצמיים נגד האנזים GAD | סוכרת מסוג 1, פגיעה בתאי בטא, תופעות נוירולוגיות |
| VDR מופחת או פגום בכליה | קולטן ויטמין D; ביטוי מופחת משנה רגישות לקלציטריול | פירוק מוגבר של ויטמין D, ייצור לא מספיק של 1,25(OH)₂D |
| RhoA / Rho‑kinase | מסלול מרכזי בהתכווצות שריר חלקי בכלי הדם | יתר לחץ דם, טרשת, איסכמיה, אי-ספיקת לב, שבץ, אי-ספיקת כליות |
| TGF-β (במצב מופרע) | בסביבה משובשת עם oxidants ממזון תעשייתי, בשר שמייצר TMAO, ביצים | דה-דיפרנציאציה, פיברוזיס, יצירת תאי גזע סרטניים |
מסלולים שמקדמים גמישות🤸, ניקוי🧹, בנייה 🏗️ושיקום🛠️. מסלולים שמאפשרים לקדם החלמה וגמישות
| ציר | מה הוא עושה | התוצאה |
|---|---|---|
| ארגינין 🫛→ NO → הרחבת כלי דם | הארגינין מומר ל-NO באנדוטליום; NO מרחיב כלי דם | ירידה בלחץ דם, שיפור זרימה, גמישות עורקים |
| ברדיקינין 🍍 | מרחיב כלי דם, מייצר NO ופרוקסיקלין | ירידה בלחץ דם, הגנה על האנדותל |
| Nrf2 🥦 | ציר הגנה מפני חמצון ודלקת | הגנה על תאי האנדוטליום, שיקום כלי דם |
| קוורצטין (בצל אדום🧅, תפוחים🍏) → Nrf2 & SIRT1 | קוורצטין מפעיל Nrf2 וסירטואינים, מפחית NF‑κB, משפר ייצור NO, מגן מפני חמצון ודלקת | הפחתת דלקת, שיפור תפקוד אנדוטלי, גמישות עורקים, הגנה על כלי דם וכליות |
| AMPK 🌦️ | מנגנון מטבולי שמפעיל פירוק שומן, מייצר אנרגיה | הפחתת דלקת, נגדי mTOR מוגבר |
| Klotho 💦 | חלבון שמעכב הזדקנות תאית | תמרון בתפקוד כליות וכלי דם, גמישות |
| רסברטרול (קמח גרעיני ענבים 🍇) → SIRT1 | רסברטרול מפעיל את האנזים SIRT1, שמאט הזדקנות תאית, מפחית דלקת, משפר מטבוליזם ומגן מפני לחץ חמצוני | עיכוב הזדקנות, הפחתת דלקת, שיפור מטבוליזם, הגנה על תאי דם וכליות, תמיכה בגמישות עורקים |
| חשיפה מבוקרת לשמש 🌞 | תומכת בייצור ויטמין D, קלציטריול | תמיכה ב-Klotho, טלומרים, שיקום |
| שיבולת שועל 🌾, קמח קליפות אננס, קטניות, אגוזים, זרעים | מספקים סיבים, ארגינין, מגנזיום, אבץ, נוגדי חמצון | ייצור NO, ירידה ב-TMAO, שיפור רגישות לאינסולין |
| TGF-β (במצב מאוזן) | בסביבה רגועה פיזיולוגית (תזונה נבונה, שמש, ברוקולי 🥦, שקט) | תיקון רב עוצמה, ריפוי פצעים, איזון חיסוני, שמירה על מבנה רקמות |
צירים נוספים: מוח, כבד, מעי, מיטוכונדריה
| ציר | מה הוא עושה | התוצאה במצב מאוזן | התוצאה במצב מופרע |
|---|---|---|---|
| ציר המעי-מוח | תקשורת דו-כיוונית בין מעי למוח דרך עצבים, הורמונים, חיסון | ויסות מצב רוח, אנרגיה, ריכוז, ייצור סרוטונין | דלקת, עייפות, חרדה, דיכאון, קשיי ריכוז |
| ציר הכבד-כליות | כבד מייצר חלבונים, מטבוליזם, פירוק רעלים; כליות מסננות ומפנות | איזון מטבולי, ניקוי רעלים יעיל, סינון כלייתי תקין | כבד שומני, חוסר איזון מטבולי, ירידה ב-GFR |
| ציר המיקרוביום-חיסון | חיידקי מעי מייצרים חומרים שמשפיעים על חיסון ודלקת | מערכת חיסון מאוזנת, דלקת נמוכה, סינון יעיל | דלקת כרונית, תגובות אוטואימוניות, מעי דולף |
| ציר המיטוכונדריה-אנרגיה | מיטוכונדריה מייצרות אנרגיה, משתתפות במטבוליזם | אנרגיה תקינה, מטבוליזם יעיל, פחות חמצון | עייפות, מטבוליזם איטי, לחץ חמצוני גבוה |
| ציר עצב הואגוס | עצב ראשי שמעביר מסרים ממוח למעי ולהפך | רוגע, עיכול תקין, איזון לב-כלי דם | לחץ כרוני, דפיקות לב, עיכול לקוי, דלקת |
הערה: המידע המובא כאן בכל המאמר ובכל הבלוג הוא כללי בלבד, חומר למחשבה ואינו מהווה ייעוץ רפואי.
אם יש לך שאלות בנוגע למה כדאי לך לעשות למען בריאותך, יש לפנות לרופא, לרשותו תקנון רפואי, ורשימת תרופות עדכנית.
המטרה הטיפולית היא:
- להפחית פעילות של מסלולי הנזק: RAAS, NF‑κB, NLRP3, TMAO, eNOS uncoupling, anti-GAD, VDR פגום, RhoA/Rho‑kinase, TGF-β מופרע.
- להגביר פעילות של מסלולי ההחלמה: ארגינין/NO, ברדיקינין, Nrf2, AMPK, Klotho, חשיפה לשמש, תזונה עשירה בסיבים וארגינין, TGF-β מאוזן.
- לתמוך בצירים נוספים: מעי-מוח, כבד-כליות, מיקרוביום-חיסון, מיטוכונדריה-אנרגיה, עצב הואגוס.
כך, הגוף מקבל הזדמנות לחזור למצב של גמישות צנרת, ניקוי, בנייה ושיקום — במקום לשקוע במצב של קשיחות, דלקת וחסימה.
זהו שינוי כיוון: לא רק "ניהול מחלה", אלא שיקום צירי החלמה ביולוגיים רדומים.
המשך בחלק ב'
חלק ב' גמישות העורקים, מה אפשר לעשות
נשארו לך שאלות
🔬אשמח להשיב על כל שאלה
בבקשה לא להתקשר משום שזה פשוט לא מאפשר לי לעבוד – אנא השתמשו באמצעים שלפניכם
למען הסר ספק, חובת התייעצות עם רופא (המכיר לפרטים את מצבו הבריאותי הכללי של כל מטופל או שלך) לפני שימוש בכל תכשיר, מאכל, תמצית או ביצוע כל תרגיל. ירון מרגולין הוא רקדן ומבית המחול שלו בירושלים פרצה התורה כאשר נחשפה שיטת המחול שלו כבעלת יכולת מדהימה, באמצע שנות ה – 80 לרפא סרטן. המידע באתר של ירון מרגולין או באתר "לחיצות ההחלמה" (בפיסבוק או YARONMARGOLIN.COM ), במאמר הנ"ל ובמאמרים של ירון מרגולין הם חומר למחשבה – פילוסופיה לא המלצה ולא הנחייה לציבור להשתמש או לחדול מלהשתמש בתרופות – אין במידע באתר זה או בכל אחד מהמאמרים תחליף להיוועצות עם מומחה מוכר המכיר לפרטים את מצבו הבריאותי הכללי שלך ושל משפחתך. מומלץ תמיד להתייעץ עם רופא מוסמך או רוקח בכל הנוגע בכאב, הרגשה רעה או למטרות ואופן השימוש, במזונות, משחות, תמציות ואפילו בתרגילים, או בתכשירים אחרים שנזכרים כאן.
For the avoidance of doubt, consult a physician (who knows in detail the general health of each patient or yours) before using any medicine, food, extract or any exercise. The information on Yaron Margolin's website or the "Healing Presses" website (on Facebook or YARONMARGOLIN.COM), in the above article and in Yaron Margolin's articles are material for thought – philosophy neither recommendation nor public guidance to use or cease to use drugs – no information on this site or anyone You should always consult with a qualified physician or pharmacist regarding pain, bad feeling, or goals and how to use foods, ointments, extracts and even exercises, or other remedies that are mentioned as such.
רשימת מקורות
שנתמכת על מחקרים עם קישורים פעילים – לפי נושאים, עם תמיכה מחקרית לקישורים וקישורים חיים (באנגלית, כי זה המקור המדעי הראשי).
הרשימה מתייחסת לכל ציר, תוסף והמלצה שכתבנו במאמר.
מקור1, מקור2
ביבליוגרפיה
מקורות מחקריים
1. קשיחות עורקים, מחלות לב וכליות
- Role of arterial stiffness in cardiovascular disease
Cecelja M, Chowienczyk P. (2012). PMC / NIH [מקור] - Arterial stiffness and coronary artery disease
Ikonomidis I et al. (2015). PubMed [מקור]. - Arterial stiffness and vascular aging: mechanisms, prevention, and therapy
Herzog MJ et al. (2025). Nature (Signal Transduction and Targeted Therapy) [מקור]. - Arterial stiffness in chronic kidney disease
(סקירה מקיפה) [מקור]. - Arterial stiffness and subsequent incidence of CKD and kidney outcomes
ScienceDirect (מחקר ארוך טווח).
[מקור].
2. RAAS, דלקת ומנגנונים מולקולריים
- Signaling pathways of chronic kidney diseases (כולל RAAS, NLRP3, NF-κB, Nrf2)
PubMed / Nature Communications.
[מקור]. - Role of Nrf2/HO-1, TLR4/NF-κB/NLRP3/caspase-1 and Wnt/β-catenin pathways
ScienceDirect [מקור]. - Targeting NLRP3 Inflammasome With Nrf2 Inducers in Central Nervous System Disorders
Frontiers in Immunology [מקור].
3. TMAO, בשר וסיכון קרדיווסקולרי
- Gut microbiota-generated TMAO and cardiovascular disease
Nature Reviews / מחקרים מרכזיים (סקירה קלאסית).
[מקור]. (Tang et al., 2014) - TMAO and risk of cardiovascular events: a systematic review and meta-analysis
Qi J et al. (2017). PubMed [מקור]
4. ארגינין, NO ותפקוד כלי דם
- L-Arginine therapy in cardiovascular pathologies (סקירה)
Böger RH. (2008). PubMed [מקור]. - Arginine and Endothelial Function (סקירה שיטתית)
Gambardella J et al. (2020) [מקור].
5. ברדיקינין ו-ACE inhibitors
- Role of bradykinin in mediating vascular effects of ACE inhibitors
Hornig B et al. (1997). PubMed [מקור]. - Bradykinin-mediated cardiovascular protective actions of ACE inhibitors
Remme WJ. (1997). PubMed.
[מקור]
6. Nrf2, חמצון ודלקת
- Nrf2 as a therapeutic target in cardiovascular disease
Li J et al. (2009). PubMed [מקור]. - Nrf2 and oxidative stress in kidney disease (סקירות מרובות)
PubMed – חיפוש מומלץ: "Nrf2 kidney oxidative stress" [מקור]
7. SIRT1, רסברטרול והזדקנות
- SIRT1 in cardiovascular aging
Luo XY et al. (2014). PubMed.
[מקור] - The protective role of Sirt1 in vascular tissue (סקירה) [מקור].
8. Klotho, ויטמין D וטלומרים
- Klotho: a potential therapeutic target in aging and cardiovascular disease
PMC / NIH. [מקור] - The relationship between vitamin D and telomere/telomerase
ScienceDirect.
[מקור]. - Vitamin D supplements may slow biological aging (2025)
Harvard Gazette [מקור]. - Daily vitamin D may slow biological aging (2026)
Futura-Sciences [מקור]
9. איריסין, PGC-1α, מיטוכונדריה ופעילות גופנית
23–24. Irisin, PGC-1α and mitochondrial biogenesis – סקירות מומלצות ב-PubMed:
"irisin PGC-1α exercise" ו- "PGC-1α mitochondrial biogenesis" [מקור1, מקור2].
10. אוטופגיה, צום והתחדשות תאית
25–26. Autophagy in aging and disease + Intermittent fasting and autophagy – סקירות מומלצות ב-PubMed [מקור1, מקור2].
11. קוורצטין, בצל אדום ו-Nrf2/SIRT1
- Quercetin activates Nrf2 pathway (מחקרים עדכניים) [מקור].
- Quercetin and SIRT1/Nrf2 activation – סקירות ב-PubMed.
12. ברומליין, דלקת ועיכול
- Bromelain: a review of its mechanisms and pharmacological effects (2023)
RSC [מקור]. - Properties and therapeutic application of bromelain
PMC / NIH [מקור].
13. CoQ10, הומוציסטאין, מתילציה
- Daily supplementation of folic acid, B vitamins, vitamin D, and CoQ10
Natural Health Research [מקור]
nta/ - Homocysteine, methylation and cardiovascular disease – סקירות ב-PubMed.
14. תרופות וצירי מחלה
- Statins and CoQ10 depletion – סקירות ב-PubMed.
- Beta-blockers and mitochondrial function – סקירות ב-PubMed.
- PPIs and microbiome / nutrient absorption – סקירות ב-PubMed.
מאמרים אחרונים
- גמישות העורקים. חלק א' על צירי המחלה מול צירי ההחלמה – הסיפור האמיתי של כלי הדם
- חלק ב' גמישות העורקים, מה אפשר לעשות
- בשר, כליות וזרימת דם מואצת: כיצד תזונה מהחי פוגעת במערכת הסינון של הגוף
- מכתב תודה ממחלימת כליות בתוך ארבעה חודשים.
- ירידה בתפקוד הכליות – מה לעשות?
- ציר פה- כליות – פגיעה בבריאות הפה בקרב חולי כליות ו-CKD
- גלוטתיון – מנוע התיקון המרכזי של הגוף
- גלוטמין (Gln) -המגן הגדול על בריאות האדם – כל מה שחשוב לדעת
- מלח שולחן ויתר לחץ דם
- תאי T בזמן החלמה: כיצד מערכת החיסון מתכיילת בלילה כדי לרפא ולא רק להילחם בגורמים למחלה
- תפקוד הכליה בשעות הלילה: השפעת המקצב הצירקדי על הפילטר הביולוגי
- מסלול איתות חדש במוח שמווסת אכילה מופרזת
- התהליכים הביוכימיים של יצירת תאי השומן בגוף, ויסות ההורמונלי והשפעות של שינוי תזונה, תוספי מזון והתעמלות על צמצומם כמות השומן באדיפוציטים על-מנת לקדם תהליכי תמיכה בתהליכי התאוששות.
- עליה במשקל ומחלת הכליות השומניות – דרכו של השומן מתאי השומן (אדיפוציטים) רוויים אל תוך איבר הכליות
- סיבי תזונה
- קמח קליפות בצל ותה קליפות בצל – מתכון
- בני-על – האם יש גבול ביולוגי למספר השנים שאדם יכול לחיות?
- 8 חסרים תזונתיים שכיחים
- האם בוטנים, קליפות בוטנים ומוצריהן הם מזון על?
- להתסיס שיבולת שועל – מתכון וכל הסודות.
- תוסף מזון על האסטקסנטין astaxanthin
- סלניום Se התגלה כאנטי- אייג'ינג ומגן מפני מחלות כרוניות
- רוצה להפעיל מנגנוני שיקום טבעיים בגוף האדם ללא תרופות ממחלת מחלת כליות נפרופתיה אימונוגלובולין A?
- משחה צהובה – לכאבי שרירים – משחת הפלא להפחתת כאבים
- מזון כתרופה
- ממצאים חדשניים למקור הגאוט
- צרבת כרונית – רוצה להפעיל מנגנוני שיקום טבעיים בגוף האדם ללא תרופות?
- מהי תסמונת מטבולית (MetS)?
- הפסיכולוגיה הטיפולית וההוליסטית – תולדות הפסיכותרפיה
- הגיל השלישי, תאוותיה של הזיקנה – ואיך להתבגר יפה, טוב ובריא
- בני-על – האם יש גבול ביולוגי למספר השנים שאדם יכול לחיות?
- כל מה שאתה צריך לדעת על מיקרו-תזונה – ויטמינים ומינרלים
- ציר המעיים-מוח פסיכוביוטיקה psychobiotics
- פאראצטמול (PARACETAMOL) גם פרצטמול או אצטאמינופן (Acetaminophen) – הידוע גם בשם אקמול, רוקסט ודקסמול עלול לפגוע בכליות ובכבד ואסור לשימוש לחולי כליה
- התרופות והרע – לחץ דם
- מקצבים בעולם החי – שעון ביולוגי בתאי הגוף
- מזון למוח – המזון הבריא למוח תומך בגמישות מערכת העצבים שלו וביכולת הלמידה, שומר על הזכרון, ומונע מחלות כגון אלצהיימר.
- מרחבי חיים מאריכי חיים – האזורי הכחולים
- אבחון מוקדם למחלת הכליות יכול להציל חיי אדם
- פורינים חוץ תאיים ותחזוקת הכליות
- פחד גבהים – מהו פחד גבהים וטיפול
- חומצת שתן נמוכה, או היפואוריצמיה (Hypouricemia)
- התערבויות בניהול רגשות, מחשבות ואורח חיים כדי להתמודד עם מגיפה של מחלות כליתיות
- הקשר בין כליה לבריחת סידן – תפקוד לא מוערך של הכליה במניעת אוסטאופורוזיס
- עשרת המזונות הבריאים ביותר לחולי כלייה – קמח קליפת רימונים (PPF) .9
- על השעורה
- עשרת המזונות הבריאים ביותר לחולי כלייה על האצות והפוקוקסנטין, חלק 7.
- עשרת המזונות הבריאים ביותר לחולי כלייה – שיבולת השועל וה β- גלוקנים חלק – 6.
- עשרת המזונות הבריאים ביותר לחולי כלייה – צרור נבטי הברוקולי והסולפורפאן חלק – 5.
- עשרת המזונות הבריאים ביותר לחולי כלייה – קסמי הכוסמת – המטפלת של דפנותה העורקים – 4
- עשרת המזונות הבריאים ביותר לחולי כלייה – תפוח האדמה והבוטיראט. 3.
- עשרת המזונות הבריאים ביותר לחולי כלייה – החסה
- עשרת המזונות הבריאים ביותר לחולי כלייה – הבצל והפלפלים. 1
- עורקים גמישים – הסוד והדרך לזכייה בבריאות מחדש
- חולשת שרירים וירידה בתפקוד השרירים. כאבי שרירים, אבדן שריר, אבדן מסת שריר ומיופתיה.
- ההומוציסטאין – האויב הגדול של האנדותל ובריאות העורקים
- ירידה בתפקוד הכליות – מה לעשות?
- על ההשפעה האיומה של חלבון מן החי על הכליות
- אילוף הכליות הסוררות
- רוצה לשנות מהלכים במחלת מחלת כליות נפרופתיה אימונוגלובולין איי?
- מחלת כליות פוליציסטית אוטוזומלית דומיננטית (ADPKD)
- דלקת כליות אינטרסטיציאלית – השתמשו במי שעורה
- התערבויות בניהול רגשות, מחשבות ואורח חיים כדי להתמודד עם מגיפה של מחלות כליתיות
- שכיחות של דיכאון וחרדה בקרב חולי
- ציר המעיים-מוח פסיכוביוטיקה psychobiotics.
- אלרגיה קשה למזון – להפעיל מנגנוני שיקום טבעיים בגוף האדם במצבי פגיעה ברירית המעיים ללא תרופות – מצרבת, השמנת יתר, אנמיה – מאבדן גמישות צנרת העיכול
- המסלולים הביולוגיים שמשקמים את הגוף – חלק ב', על הפוטנציאל הקיים בגוף האדם לקדם תהליכי החלמה מאת ירון מרגולין
- המסלולים הביולוגיים שמשקמים את הגוף – חלק ג', על התקדמותם של צירי ההחלמה בגוף האדם
- 🔷 המסלולים הביולוגיים שמשקמים את הגוף: p53 — שומר הגנום – חלק ד’ — ירון מרגולין
- חלק ה' – חלבוני ההחלמה: הרשת המולקולרית של הריפוי, ההתחדשות וההומאוסטזיס מאת ירון מרגולין
- חלק ו' על צירי ההחלמה ומה שמפעיל אותם – (signal architecture) מאת ירון מרגולין
- מנגנוני תיקון פנימיים בגוף: מודל אינטגרטיבי להבנת תפקוד הכליות והמערכת כולה – 3 צירי ההחלמה המרכזיים
- המסלולים הביולוגיים המשקמים את הגוף: פרספקטיבה חדשה על החלמה
- ⭐ התחדשות כלייתית: מקרה נדיר של ריפוי טבעי — ומה שהוא מלמד את כולנו
- דרכים חדשות לוויסות יעיל של טריגליצרידים (Triacylglycerols) בדם
- אי ספיקת כליות – טיפול ללא תרופות
- על ההחלמה – למה הגוף לא מחלים — וכיצד להחזיר לו את היכולת הטבעית לעשות זאת
- שינה חשובה להחלמה – 🌜זמן הריפוי החשוב מתרחש בלילה
- מיומטי תנועה – התקווה החדשה לחולי כליות: תרכובות שמחקות את צירי ההחלמה של פעילות גופנית
- להפעיל מנגנוני שיקום טבעיים בגוף האדם בפשטות ממחלת ריאות חסימתית כרונית
- רוצה להפעיל מנגנוני שיקום טבעיים בגוף האדם בסוכרת
- שחיקת סחוס, למה לסבול? –
- עורקים גמישים – הסוד והדרך לזכייה בבריאות מחדש
- פרנצ'סקו מסינה Francesco Messina אלהים באדם שחיפש אחר היופי
- התרופות הטובות והרע – חיסול האוצר המגן על משק המיים ולחץ הדם בגוף – האשלגן
- התרופות הטובות והרע – אבץ
- תרופות הרגעה שמשאירות אותך רעב וחרד – ציפּרָלֵקס, פּרוֹזַק, פלואוקסטין
- התרופות והרע – ביוטין B7 ויטמין H
- רוצה להפעיל מנגנוני שיקום טבעיים בגוף האדם ביתר לחץ דם?
- התרופות והרע – ומי הורס לך את ה-Q10
- מחסור בויטמין בי-1 מייצר סיוטי לילה וחלומות זוועה B1
- גלוטתיון Glutathione ו-N-אצטיל-ציסטאין (NAC)
- למה יש לי מחסור בוויטמין B12
- אלרגיה קשה למזון – להפעיל מנגנוני שיקום טבעיים בגוף האדם במצבי פגיעה ברירית המעיים ללא תרופות – מצרבת, השמנת יתר, אנמיה – מאבדן גמישות צנרת העיכול
- אבנים בכליות – אוקסלטים, היפרוקסלוריה ויתר חומצה אוקסלית – רוצה להפעיל מנגנוני שיקום טבעיים בגוף האדם ללא תרופות?
- זרימת דם כלייתית
- השפעות של הורמון הגדילה (GH) על תפקוד הכליות בבריאות ובמחלות כליות
- סמן ביולוגי אמין (ביו-מרקר)
- למאמרי התכנית לשיקום הכליות – כאן
- המסלולים הביולוגיים שמשקמים את הגוף – חלק ב', על הפוטנציאל הקיים בגוף האדם לקדם תהליכי החלמה מאת ירון מרגולין
- סמנים ביולוגיים (Novel Biomarkers) ובדיקת כליות מוקדמת ככל האפשר – מצילה חיים
- אבחון מוקדם למחלת הכליות יכול להציל חיי אדם
- אי ספיקת כליות
- PGC-1α, יעד טיפולי חדש נגד מחלת הכליות
- התערבויות בניהול רגשות, מחשבות ואורח חיים כדי להתמודד עם מגיפה של מחלות כליתיות
- פורינים חוץ תאיים ותחזוקת הכליות
- רככת היפופוספטמית (זרחן נמוך מהנורמה) – מחלת כליות כרונית – או הפרעת עצם מינרלית
- סלניום Se התגלה כאנטי- אייג'ינג ומגן מפני מחלות כרוניות
- מלטונין הוכח שהורמון השינה נוגד חמצון רב עוצמה ויעיל כתרופה בקרב חולי כליה
- רעלים אורמיים מקדמים דיאליזה – הוכח לאחרונה שרעלנים אורמיים קשורים למיקרוביוטה של המעי הגס – איך להחלים ללא תרופות ולהימנע מדיאליזה
- מאמר הצלבת איברים – הדרך להבנת המחלה– כאן.
- מיטוכונדריה – במיטוכונדריון מתחילה מחלת הכליות
- תובנות חדשות אי-ספיקת כליות תלויה במצבו של ציר FGF23-Klotho
- מחלת שינוי מינימלי Minimal Changes Disease מינימל צ’יינג’
- מחלת כליות פוליציסטית אוטוזומלית דומיננטית (ADPKD)
- התכנית לשיקום הכליות – כאן.
- מכתב תודה ממחלימת כליות בתוך ארבעה חודשים.
- המזון הבסיסי והחומצי – מה כן ומה לא נכון. מה עובד על שיקום תפקודי, ומה הן אגדות? חומציות מגיעה מתזונה והיא קמה והורסת את הכליות
- עליה במשקל ומחלת הכליות השומניות – דרכו של השומן מתאי השומן (אדיפוציטים) רוויים אל תוך איבר הכליות
- הקשר של כבד שומני, ומחלת כליות כרונית – מחקר חדש.
- מחלת כליות – תמיכה בתהליכי התאוששות – לשם כך יש לזהות אותה מוקדם ככל האפשר – הקסטסרופה!
- זיהום סביבתי מקדם את מחלת הכליות
- מחלת כליות פוליציסטית אוטוזומלית דומיננטית (ADPKD)
- השפעות של הורמון הגדילה (GH) על תפקוד הכליות בבריאות ובמחלות כליות
- כלית העל מספרית Accessory kidney
- דנרבציה כלייתית Renal denervation (RDN)
- השפעות של הורמון הגדילה (GH) על תפקוד הכליות בבריאות ובמחלות כליות
- אנמיה למה חסר לי ברזל?
- פגיעה במערכת העיכול ללא תרופות?
- קל לשמור על הכבד
- להחלים בפשטות ממחלת ריאות חסימתית כרונית
- סוכרת
- שחיקת סחוס, למה לסבול? – תמיכה בתהליכי התאוששות
- עורקים גמישים – הסוד והדרך לזכייה בבריאות מחדש
- פרנצ'סקו מסינה Francesco Messina אלהים באדם שחיפש אחר היופי
- התרופות הטובות והרע – חיסול האוצר המגן על משק המיים ולחץ הדם בגוף – האשלגן
- התרופות הטובות והרע – אבץ
- תרופות הרגעה שמשאירות אותך רעב וחרד – ציפּרָלֵקס, פּרוֹזַק, פלואוקסטין
- התרופות והרע – ביוטין B7 ויטמין H
- יתר לחץ דם
- התרופות והרע – ומי הורס לך את ה-Q10
- מחסור בויטמין בי-1 מייצר סיוטי לילה וחלומות זוועה B1
- גלוטתיון Glutathione ו-N-אצטיל-ציסטאין (NAC)
- הגוף כמעבדה: ריפוי תאי באמצעות ידע, תודעה מדויקת וממוקדת בצרכים הפיזיים שהגוף דורש להחלמתו
- התהליכים הביוכימיים של יצירת תאי השומן בגוף, ויסות ההורמונלי והשפעות של שינוי תזונה, תוספי מזון והתעמלות על צמצומם כמות השומן באדיפוציטים על-מנת לקדם תהליכי תמיכה בתהליכי התאוששות.
- שלושת המיצים
- חזרה לבסיס – עקרונות צירופי המזון
- על ההזנחה –
- ניתוק רגשי – על הקורוציונה
- די לכאב
- ראיית המעמקים – כניסה לטרקלין או על החיים האמתיים.
- התודעה השלילית
- ביקורת מבזה
- רוצה לקגם מצבי יתר לחץ דם?
- התרופות והרע – ומי הורס לך את ה-Q10
- מחסור בויטמין בי-1 מייצר סיוטי לילה וחלומות זוועה B1
- גלוטתיון Glutathione ו-N-אצטיל-ציסטאין (NAC)
- שלושת המיצים
- חזרה לבסיס – עקרונות צירופי המזון
- על ההזנחה –
- ניתוק רגשי – על הקורוציונה
- די לכאב
- נאטו (פולי סויה מותססים יפניים) מתכון
- הקשר של כבד שומני, ומחלת כליות כרונית – מחקר חדש.
- טאורין: חומצה אמינית מסתורית ומסקרנת
- מלטונין הוכח שהורמון השינה נוגד חמצון רב עוצמה ויעיל כתרופה בקרב חולי כליה
- דיוקסינים במזון