ירון מרגולין
הכבד השומני אינו רק מאגר של שומן. הוא משקף שינוי עמוק בהחלטות המטבוליות של התא: המתבונן בעיני המחקר מבחין מעבר ממסלולי שריפה וייצור אנרגיה למסלולי אגירה… מבט אל רפואת העתיד
הכבד הוא איבר מרכזי שבו משתקפים עומס מטבולי, דלקת, עקה חמצונית, שיבוש תזונתי, פגיעה בגלוקוז ובשומנים, ולעיתים גם ירידה תפקודית שאינה תמיד שקולה לנזק מבני בלתי הפיך.
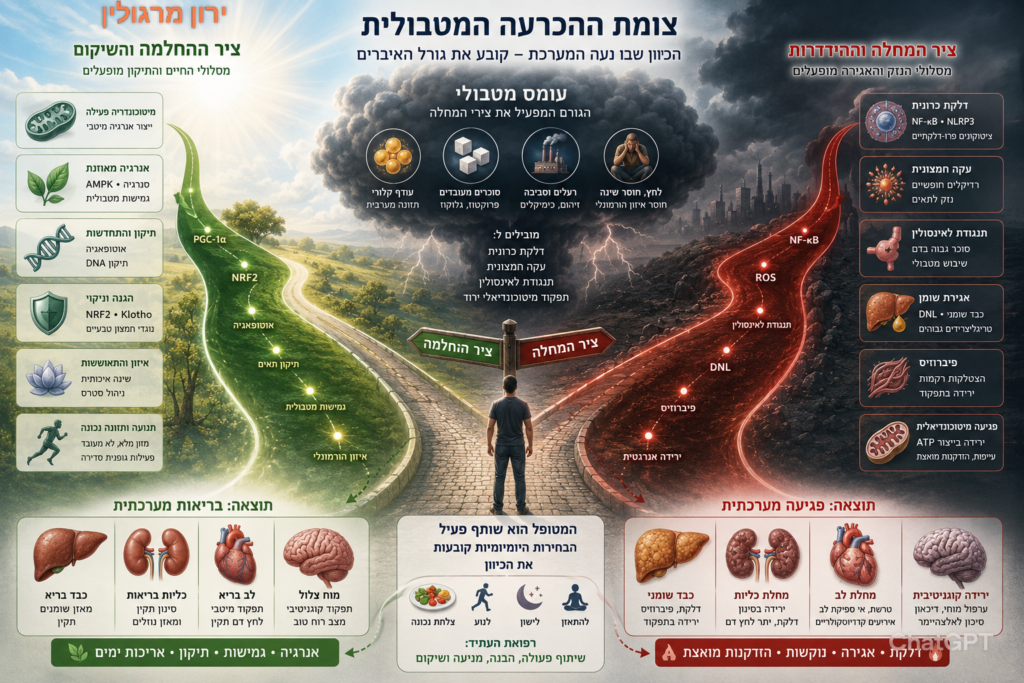
הכבד כ"שופט" או "מאזניים" המכריעים בין שריפה לאגירה — בין מחלה ותפקוד מיטבי מוצג בציור למעלה. איור שמשקף את העמדה החדשה שצומחת מהמחקר ומהתבוננותם של מבקשי המחלה, רפואת העתיד:
הכבד כבר לא נראה כקורבן של השומן. הכבד הוא אבר מכריע.
במיתולוגיה המצרית הלב נשקל מול נוצת האמת, וההכרעה קובעת את גורל האדם. במאמר שלפניך ההכרעה אינה מוסרית אלא מטבולית:
- האם הפחמן ינותב למיטוכונדריה?
- האם יישמר כגליקוגן?
- האם יהפוך לשומן?
רעיון שפותח חלון לקורא ודלת למטפלים החדשים, הצעירים, דור ההייטק שמשתקף במחקרים ומפתח לאורך כל סדרת "צירי ההחלמה והמחלה", מסלולים ביולוגיים רדומים, שהתעוררותם מקדמת החלמה – 3 הצירים המרכזים הוצגו במבוא או מאמר כללי שפתח את הסדרה:
- העתיד – חלק א'
- עורקים גמישים חלק ב', הסבר מפורט על הצירים. הביולוגיה העמוקה של גמישות העורקים מאת ירון מרגולין
- מסלולי ההחלמה -המסלולים הביולוגיים שמשקמים את הגוף – חלק א' מאת ירון מרגולין
לא חומר בודד קובע את הגורל, לא מבט מנותק בפסגה, אלא ראיית המסע אל ההר, ההכנות, והטיפוס על הקשיים ומזג האוויר, מבט אל כיוון הזרימה בצומת ההכרעה, וצומת זו במקרה שלפנינו הינה הכבד:
"הכבד: שופט האנרגיה של הגוף"
או אפילו:
"הכבד השומני: סיפורו של שופט שאיבד את האיזון"
או:
"צומת ההכרעה: מתי הכבד שורף ומתי הוא אוגר?"
ציור שמשקף ידע חדש, שנישען על הקיים, לא עוד האין כמשענת: הוא אינו מציג את הכבד כמכונה ביוכימית בלבד, אלא כישות שמקבלת החלטות. כמובן שמדעית אין "החלטה", אבל כמשל אפלטוני, למעשה זה מדויק להפליא, משום שכל המאמר עוסק במעבר בין מסלולים מטבוליים מתחרים. ומציג לפניך הן את הסנגור והן את הקטגור שמאפשרים לכבד לשפוט נכון, עד שמגיעה כמובן השחיתות הנפוצה, אבל היא לא חלק מהביולוגיה, היא למעשה המחלה.
מאמר זה אינו מציע טיפול ואינו מבטיח החלמה. הוא מציע כיוון מחשבה.
במשך שנים ניסיתי לסייע לאנשים להבין את מחלתם, אך לא פעם גיליתי שדווקא הם סייעו לי להבין טוב יותר את הגוף האנושי. ואני מודה להם.
הדפים הבאים הם חלק מאותו מסע לימוד משותף.
בעיניי, זה משקף היטב את הרוח שעולה
הקדמה
לפני 20 שנה כתבתי על "ניקוי כבד" "קל לנקות את הכבד" כתהליך נקודתי.
היום אני מבין שהכבד אינו רק איבר שלעיתים הוא חולה וספוג שומן — אלא הוא איבר שמשקף את מצב המערכת כולה. באותם ימים דרכי השיקום התענינו בעיקר בכמות השומנים שבאבר ובתזונה, צמחים והפחתת עומס.
המקור העיקרי לשומן שמצטבר בכבד ידוע כיום. הוא אינו שומן מהמזון, אלא עודף פחמימות וסוכרים (במיוחד סוכר פשוט ופרוקטוז) שמגיע מהמזון.
מנגנון הפעולה בגוף בו מצטבר שומן בכבד עובד בצורה הבאה:
1. ייצור שומן חדש (De Novo Lipogenesis)
כאשר אוכלים פחמימות🥐 וסוכרים🍨 בכמות העולה על צורכי האנרגיה המיידיים של הגוף, מאגרי הגליקוגן בכבד מלאים, עודפי פחמימות (ובמיוחד פרוקטוז וגלוקוז) מאלצים את הכבד לעבד את העודפים הללו, כדי לסלקם ממנו. אז הם עוברים מסלול ביוכימי שמפרק אותם ל-Acetyl-CoA, אשר הופך בהמשך לחומצות שומן רוויות ולטריגליצרידים בתהליך ה-De Novo Lipogenesis (DNL). תהליך כימי שנקרא "ייצור שומן מחדש" (De Novo Lipogenesis) [מקור]. השומן הזה נאגר ישירות בתוך תאי הכבד (גול עצמי). מדובר באחד הצירים המטבוליים המרכזיים בהתפתחות כבד שומני.
גול עצמי
הפרדוקס הביולוגי והפיזיולוגי של הגוף המודרני ניצב לפנינו.
המבט אל הציר אשר בקיצור הוא מכונה DNL [ מקור1, מקור2] נוכח כבד שומני מציף תחושה של "אבסורד", היא נובעת מהבנת המסלול, ומכך שהידע חושף פער עצום בין הסביבה שבה הגוף שלנו מתפקד היטב (אולי זו סביבה אבולוציונית של מחסור במזון) לבין הסביבה שבה אנו חיים היום (שפע קיצוני של פחמימות זמינות) [מקור1, מקור2].
כדי להבין מדוע הכבד עושה משהו שנראה כמו "גול עצמי", צריך להסתכל על התהליך משתי נקודות מבט:
הטווח הקצר (הישרדות מיידית) והטווח הארוך (שכולל את המחיר הבריאותי).
הכבד לא מנסה להזיק לעצמו בכוונה. הוא פועל לפי חוקי הישרדות עתיקים:
עדיף כבד שומני בעוד כמה שנים, מאשר מוות מרעילות סוכר בדם בעוד כמה שעות. כאן נכנסת לתמונה דמותו כשופט. והשיפוט נובע מאחריות אמיתית.
המנגנון של De Novo Lipogenesis (DNL) הופך נוכח שפע תזוני (לחם, אורז, איטריות, תפוחי אדמה, קרואסון, עוגות, עוגיות, קרקרים…) את הכבד ממווסת מטבולי למפעל שמייצר שומן מחדש [מקור1, מקור2]. פרוקטוז מאיץ את התהליך הזה משום שהוא עוקף חלק ממנגנוני הבקרה המטבוליים, מוזרם ברובו לכבד, ומפעיל מסלולים ליפוגניים במהירות גבוהה יותר המהלך מורכב יותר ויובא בהמשך, אבל מה שניכר שמדובר במנגנון מולקולרי במסלול שבו פחמימות הופכות לשומן שנאגר בכבד, אנחנו נדרשים אם כן להסתכל על המסלול המטבולי שהתא עובר,
אגירת השומן בכבד יותר נחשפת כמדד. היא רק המדד שמשקף מערכת שלמה שנכנסת לגול עצמי.
והמסלול המטבולי שהוא אחד משלושת המסלולים הביולוגיים המשקמים את הגוף, אם הם פועלים נכון. השומן המצטבר בכבד זו השתקפות של צירים אלו.
המסלולים הביולוגיים המשקמים את🔹 הגוף: פרספקטיבה חדשה על החלמה – מבט עומק על על 3 המסלולים המרכזיים.
כיצד הגוף עובר מציר של בנייה ושיקום לציר של אגירה, דלקת ופגיעה?
כבד שומני אינו תחילתו של הסיפור. הוא אחת התחנות שאליהן מגיעה מערכת שנעה לאורך ציר מחלה. כאשר משתנה הכיוון, משתנה גם גורלו של הכבד.
כיום אנו עומדים בפתח של רפואת העתיד והדברים נראים קצת אחרת: הכבד, הכליות כבר לא עוד איבר שיש לטפל בו, וככל הנראה זה המצב לגבי המוח, אלא איבר שמגיב ומשקף את מצב המערכת כולה. לא השומנים, כמות השומנים שהיא הבעיה, אלא העומס המטבולי. וכאשר יש עומס מטבולי, דלקת, עקה חמצונית ותפקוד מיטוכונדריאלי ירוד, צירי השיקום, מסלולי ההחלמה נחסמים ותחתם קמים ופועלים ולפעמים במרץ מסלולי מחלה ודלקת, אז גם אברים מרכזיים כמו כליות, לב, וכבד נראים כמי שנפגעו [מקור1, מקור2].
ההבחנה בין הנזק הנראה לבין הגורם המפעיל אותו.
המאמר מלפני 20 שנה היה הראשון שפרסמתי כשהחלתי לעבוד כמטפל ולפני שהבלוג החל להוות יעד להתקפות, המאמר שמופיע כיום בבלוג ממאי 2008 [מקור] הוא שיחזור שלו. ומציג גישה שעדיין מענינת, וכוללת כבר את ההבחנה בין הנזק הנראה לבין הגורם המפעיל. אבל מזמן נעשתה הרפואה ההוליסטית, הטבעית, רחוקה מהמחקר וישנה. לפני עשרים, שלושים שנה הייתה זו יותר ה"רפואה טבעית" קלאסית, תחום שכיח כיום, אבל בחלקו מיושן תחום שכשל בהפעלתם של מנגנוני החלמה במחלת הכליות למשל [מקור]:
כיום אנו ניצבים בפתחו של שינוי תפיסתי ברפואה. הכבד, הכליות, הלב ואפילו המוח אינם רק איברים שנפגעו ודורשים טיפול. הם משקפים את מצבה של המערכת כולה.
כבד שומני אינו בהכרח בעיה של שומן, כפי שמחלת כליות אינה בהכרח בעיה של דלקת בכליה. פעמים רבות מדובר בביטוי מקומי של עומס מטבולי רחב יותר.
כאשר עומס מטבולי, דלקת כרונית, עקה חמצונית ופגיעה בתפקוד המיטוכונדריאלי מצטברים, מסלולי השיקום והתיקון נחסמים. במקומם מופעלים מסלולי מחלה, אגירה ודלקת. הכבד, הכליות, הלב והמוח אינם הגורם הראשוני, אלא לעיתים קרובות הראשונים לחשוף את המשבר המתחולל במערכת כולה. למעשה הופעת כשל תפקודם היא מדד למצב רחב שמתחיל פעמים רבות בצלחתם של החולים.
:בעבר
- ניקוי כבד כתהליך נקודתי
- דגש על צמחים (גדילן), תוספים (לציטין), רעלים (הפחתת פחמימות, אלכוהול)…
- תיאורים מטפוריים על כעס, כעס "תקוע", דימוי עצמי, יחודיות, מרכזי חשיבה בהשפעת הרפואה העתיקה (היפוקרטס).
- פעם אם הפעלנו צירי החלמה הייתה פחות הבנה לכך. המבט אל הצלחת היה, אבל לא אל נושא המסלולים המולקולריים, חלבוני שיקום. מבט זה חדש ומגיע מהמחקר המדעי ולא ידענו למעשה להפעיל את המסלולים הביולוגיים שמשקמים את הגוף בצורה מדויקת. אבל תוצאות המעבדה הראו שפעלנו נכון. הושגה תרופה, מזון כתרופה, צירי המחלה נחסמו ,❌. הייתה תרופה👌המטרה הושגה ודי בקלות בדרך כלל. אנשים ראו מדדי טווח נורמה, ונהנו. אבל כשמדדי טווח הנורמה החלו להופיע בקרב חולי כליה, נושא התרופה שהושגה או לא הושגה היה כתחלופה לכמיהה לציון או חזרה לגן עדן. אגדה יפה על גיזת זהב. בפועל האתר yaronmargolin.com הפך יעד להתקפות ומאמרים הופלו, ושובשו, והופלו גם נפגעו שוב ושוב מערכות האימייל בבלוג, עד היום חסר המאמר על צירופי המזון, ועל השלבים, "שלבים: משבר והתפתחות אצל היחיד" (Stages: Phases of Development in Human Life) שהציג את עיקרי הדברים מאת הסופר והפסיכיאטר ההולנדי ברנרד ליווכוד שהיה באותם זמנים מרתק ומהפכני, למעשה גם כיום.
- היום אנחנו מתבוננים על ביולוגיה מערכתית חדה יותר. לא מתמקדים בצירי מחלה, בחלבון זה או אחר בציר, שמיועד לבלימה על ידי כדור 💊. המחשבה שונה: חלבון שמקדם דלקת או פגיעה בDNA, עדיין חשוב, במיוחד במחקר, למען תעשיית התרופות, אבל סיבת הפעלתו מתרדמתו, חשובה כיום הרבה יותר. כך, המבט החל להתבונן אל המסלולים הביולוגיים המשקמים ומקדמים החלמה בגוף: אל חלבוני שיקום ואל אנזימי החלמה.
- צירי מחלה (RAAS, NF‑κB, NLRP3, TMAO, Rho‑kinase) [מקור1, מקור2], מול צירי החלמה (Nrf2, AMPK, SIRT1, אוטופגיה, Klotho)
- הבנה של כבד כמראה של מערכת, לא רק "איבר חולה", מבט אל כל המרחב.
- תפיסה של גמישות ביולוגית, הפעלת צירי החלמה וחלבוני שיקום, ולא רק "ניקוי" בולטת בנושא החזרתם של דפנות העורקים לתפקוד, גמישות העורקים – רפואת העתיד – חלק א' ומובילה בתהליכי שיקום כלייתיים – מנגנוני תיקון פנימיים בגוף: מודל אינטגרטיבי להבנת תפקוד הכליות והמערכת כולה. נושא שחדר לאחרונה לטיפול ב אנמיה, אנמיה – מחסור בברזל, והיבטים ביולוגיים חשובים של מחסור בברזל מחשבות חדשות – מחקר חדש גם אנמיה – מחסור בברזל, היבטים ביולוגיים והתבוננות על דרכי הפעלה של מנגנוני שיקום טבעיים בגוף האדם – מחקר חדש.
- מסלולי ההחלמה -המסלולים הביולוגיים שמשקמים את הגוף – חלק א' מאת ירון מרגולין.
- המסלולים הביולוגיים שמשקמים את הגוף – חלק ב', על ההחלמה מאת ירון מרגולין
- חלק ג' – על ההחלמה
- חלק ד' – על שומר החומר הגנטי🔹🐦🔥 P53 🐦🔥 והנחיות טבעיות של הגוף ל: "החלמה".
- חלק ה' – חלבוני ההחלמה: הרשת המולקולרית של הריפוי, ההתחדשות וההומאוסטזיס מאת ירון מרגולין
- חלק ו' על צירי ההחלמה ומה שמפעיל אותם – (signal architecture) מאת ירון מרגולין
- 🔹 מפת מסלולי ההחלמה – 3 צירים – מבוא או מאמר כללי
- המסלולים הביולוגיים המשקמים את🔹 הגוף: פרספקטיבה חדשה על החלמה – מבט עומק על על 3 המסלולים המרכזיים.
- גמישות העורקים – רפואת העתיד – חלק א'
- עורקים גמישים חלק ב', הסבר מפורט על הצירים. הביולוגיה העמוקה של גמישות העורקים מאת ירון מרגולין
המנגנון הביולוגי של ייצור שומן מחדש בכבד (De Novo Lipogenesis – DNL) עומד במרכז הכבד השומני, כשל ציר זה משתקף במחלת כבד, גם אם הכבד תקין.
מנגנון ה-DNL ועודף פחמימות
השפע, יותר פחמימות וסוכרים מכפי שהגוף זקוק להם, ובעיקר כאשר יש עודף אנרגיה מתמשך, העודף לא נשאר רק כגלוקוז בדם או כגליקוגן בשריר ובכבד. הוא מנותב ליצירת חומצות שומן חדשות, שמורכבות בהמשך לטריגליצרידים ונאגר בתוך תאי הכבד [מקור]. כך, הכבד לא רק “סופג” עודף אנרגיה — אלא הוא הופך למפעל שמייצר שומן מחדש [מקור1, מקור2]. אבל, מי יצר את הבעיה שמטופלת בתרופות?
החשיבות של הציר הזה היא לא רק כמותית אלא איכותית: בשינו הכיוון המטבולי של הכבד. במקום להישען על חמצון שומנים ועל איזון אנרגטי, הכבד נאנס, ועובר לעודף סינתזה של שומן ולאגירה תוך-תאית. הטרגדיה שמצב זה יכול להתקיים גם כאשר הכבד עדיין נראה “תקין” מבחינה מבנית, משום שהשינוי מתחיל הרבה קודם וברמת המטבוליזם, ורק אחר כך הוא מתבטא כפגיעה ברורה ברקמה. במילים אחרות,
DNL הוא ציר שמקדים את המחלה הנראית לעין, בגלל מה שמונח בצלחת🍚🥮🍺.
לשם הבהרה
DNL הוא מנגנון ביולוגי של יצירת שומן מחדש בכבד.
DNL הוא מסלול מטבולי טבעי, אך כאשר הוא מופעל ביתר 🥮🍚🥐🍨🍔🍰🍮🍸🍻 — בעיקר בעקבות עודף פרוקטוז ופחמימות — הוא נעשה ציר שמקדם הצטברות שומן ונזק מטבולי בכבד [מקור]. לכן, אחת המטרות המרכזיות העומדות בפני המבקשים להחזיר את הציר לתפקוד מאוזן היא להפחית את העומס שמפעיל אותו ולחזור לאיזון מטבולי.
פרוקטוז
פרוקטוז (סוכר פירות) הוא חד-סוכר טבעי המצוי בפירות, דבש וירקות, ומשמש כמרכיב בסיסי בסוכר לבן ובסירופ תירס חומר שנדחס בכמות גדולה עתיר למזון, ומשקאות בתעשיית המזון. הוא מתוק יותר מגלוקוז ומעובד ישירות בכבד.
כאשר פרוקטוז מגיע לכבד, הוא אינו נכנס לאותו מסלול בקרה עדין שבו הגוף מווסת בדרך כלל את שימוש האנרגיה שלו. במובן הזה, הוא כאילו עוקף את מערכת האיזון הטבעית, ונכנס ישירות לציר מטבולי שממהר להפוך את העודף שהוא מביא לשומן. במקום לשמור על ההומאוסטזיס — אותו איזון פנימי עדין שהגוף מנסה להחזיק כל הזמן — הכבד נדחף למסלול של ייצור שומן, אגירה והעמסת יתר.
אפשר לומר שבמצב כזה הכבד כבר לא רק “מעבד סוכר”, אלא מגיב לעומס שמסיט אותו מהתפקיד המאוזן שלו [מקור]. זו בדיוק הנקודה שבה עולה מושג איכות המזון: לא רק כמה אוכלים, אלא איזה ציר מושפע ממנו ובהקשר של הפרוקטוז לאיזה ציר מטבולי הגוף מנותב. וכשהציר הזה משתבש, מתחיל להיווצר אותו הרקע שממנו מתפתח כבד שומני.
ובכבד הפרוקטוז עשוי להפוך לשומן (טריגליצרידים) ובעת צריכה מופרזת הוא מאיץ את התהליך הזה עוד יותר. בניגוד לגלוקוז, שכניסתו למסלולי הפירוק הכבדי מווסתת יותר על ידי בקרה אנרגטית, פרוקטוז נכנס למסלול כבדי שבו הבקרה חלשה יותר, (גלוקוז עובר דרך מערכת בקרה אנרגטית עדינה: כשהתא כבר מלא באנרגיה, הגוף מאט את פירוק הגלוקוז כדי לשמור על הומאוסטזיס. לעומת זאת, פרוקטוז נכנס למסלול כבדי פחות מבוקר, ולכן הוא עלול לעקוף את מנגנון הבלימה הזה ולדחוף את הכבד לעבר ייצור שומן) ולכן הוא יכול להזרים במהירות תוצרי ביניים ליפוגניים. מאחר שרוב העיבוד שלו מתרחש בכבד, הוא מגביר יצירה של מולקולות כמו אצטיל-CoA, תומך בהפעלת גורמים ליפוגניים כמו SREBP1c ו-ChREBP, ומעודד ייצור שומן בקצב גבוה יותר. בנוסף, פרוקטוז קשור גם לעקה מיטוכונדריאלית, ירידה בחמצון חומצות שומן, יצירת ROS ועומס מטבולי נוסף.
הערה: בקרה אנרגטית פירושה שהגוף לא רק “שורף סוכר”, אלא מודד כל הזמן את מצב האנרגיה של התא ומחליט אם להמשיך לפרק גלוקוז או לעצור. תזכורת: אחרי ארוחה, הגלוקוז בדם עולה, הלבלב מפריש אינסולין, והגוף מכניס את הגלוקוז לתאים או מאחסן אותו כגליקוגן [מקור1, מקור2, מקור3]. כשיש בתא מספיק אנרגיה — כלומר יש די ATP — המערכת מאטה מסלולים שממשיכים לפרק גלוקוז, כדי לא לייצר עודף אנרגטי מיותר [מקור1, מקור2, מקור3].
למה זה חשוב למחלת הכבד
הכבד הוא אחד האיברים שמנהלים את האיזון של הציר האנרגטי בגוף האדם [מקור]. אם רמת האנרגיה בתא כבר גבוהה, הגוף מעדיף לעצור פירוק נוסף של גלוקוז ולעבור לאחסון או לשימוש מסודר, ולא להמשיך לייצר עוד תוצרי ביניים [מקור1, מקור2].
הקשר לפרוקטוז
זה בדיוק המקום שבו פרוקטוז שונה: הוא נכנס למסלול כבדי שמושפע פחות מאותה בקרה עדינה, ולכן הוא יכול “לעקוף” חלק מהבלמים שמווסתים את זרימת האנרגיה [מקור]. לכן הוא נוטה יותר לדחוף את הכבד לעבר יצירת שומן מחדש במקום לשמור על איזון אנרגטי.
פרוקטוז ועודף פחמימות מסיטים את הכבד ממסלול של איזון מטבולי למסלול של יצירת שומן.
פרוקטוז ועודף פחמימות משנים את מסלול הפעולה של הכבד: ממערכת מווסתת שמבקשת לשמור על איזון, למערכת שמופעלת לייצור ואגירת שומן.
לכן, השאלה הנשאלת כבר אינה רק כמה סוכר נכנס לגוף, אלא לאיזה מסלול מטבולי הוא מנותב. כאשר עודף פחמימות — ובמיוחד פרוקטוז — דוחף את הכבד ל-DNL, הכבד משנה את תפקידו: ממווסת מטבולי למייצר-שומן. זהו אחד הצירים החשובים ביותר בהבנת הכבד השומני, ולעיתים גם אחד המוקדמים ביותר.
ממווסת מטבולי למייצר-שומן.
זהו אחד הצירים החשובים ביותר בהבנת הכבד השומני, ולעיתים גם אחד המוקדמים ביותר.
- מקורות תזונתיים נפוצים של פרוקטוז מרוכז שנוטים לפספס?
- סוגי פעילות גופנית שמסייעים לפנות את מאגרי הגליקוגן ביעילות?
- תוספים או רכיבי תזונה שנמצאו במחקרים כמסייעים להפחתת הצטברות שומן בכבד?
פתיח קצר:
הכבד אינו עומד לבדו — הוא מציג את מה שמתרחש בכל הגוף [מקור1, מקור2, מקור3, מקור4, מקור5, מקור5, מקור7]
כאשר מסלולים כמו RAAS, NF‑κB, NLRP3 ו-TMAO פעילים לאורך זמן, הכבד נע לעבר שומן, דלקת ועקה חמצונית.
כאשר אנו תומכים ב-Nrf2, AMPK, SIRT1, אוטופגיה ו-Klotho, הכבד חוזר לתפקוד.
אתחיל עם הכבד כמראה מערכתית
- תפקודי כבד בסיסיים
- כבד שומני כמדד, לא רק מחלה
- הקשר בין כבד, מעיים, תזונה, דלקת ועקה חמצונית [מקור1, מקור2]
הכבד כמראה של מצב מערכתי.
פרק 2: צירי מחלה
- RAAS, NF‑κB, NLRP3, TMAO, Rho‑kinase
- צירי מחלה: דלקת←, שומן←, עקה חמצונית←, תנגודת לאינסולין←, עומס תרופתי, ←חמצון←, נזק תאי [מקור1, מקור2].
- השפעת סוכר, שומן רווי, אלכוהול, תרופות [מקור1, מקור2].
פרק 3: צירי החלמה
- Nrf2, AMPK, SIRT1, אוטופגיה, Klotho, NO
- צירי החלמה: תזונה מניעה, ←תנועה או אימון גופני וריקוד ←, שינה←, נשימה←, ירידה בעומס סוכר ושומן ←, הפחתת עומס נפשי ← ותמיכה במנגנוני תיקון.
- תמיכה במנגנוני תיקון טבעיים [מקור1, מקור2].
נסיים ב
פרק 4: מה עושים בפועל
- תזונה: ירידה בסוכר, שומן איכותי, חלבון מספק, שומני אומגה-3
- תוספים: גדלין, כולין, הלצטין, קו-אנזים Q10, ויטמיני B
- תנועה, שינה, נשימה, הפחתת עומס נפשי
- מניעה ולא רק טיפול
ביולוגיה מערכתית שמשקפת ומקדמת מחשב והתבוננות על:
צירי מחלה (RAAS, NF‑κB, NLRP3, TMAO, Rho‑kinase) מול צירי החלמה [מקור1, מקור2] (Nrf2, AMPK, SIRT1, אוטופגיה, Klotho) [מקור1, מקור2].
הבנה של כבד כמראה של מערכת, לא רק "איבר חולה"
תפיסה של גמישות ביולוגית ולא רק "ניקוי" ← בסוף המאמר
יהיה מעניין
קריאה מהנה
ירון מרגולין
הכבד כמראה מערכתית: צירי מחלה וצירי החלמה
פתיח
המשמעות של הגישה החדשה עמוקה. במקום להסתכל על הכבד כעל “בעיה מקומית”, כדאי לראות בו צומת ביולוגית: הוא מגיב למסלולים דלקתיים, מטבוליים ומיטוכונדריאליים, והוא גם מסוגל להשתקם כאשר התנאים משתנים. מסלולים כמו NLRP3, NF-κB ועקה חמצונית דוחפים לעבר שומן, דלקת ופיברוזיס; לעומתם, Nrf2, AMPK, SIRT1, אוטופגיה ומיטופגיה תומכים בניקוי, תיקון וחזרה לתפקוד [מקור1, מקור2, מקור3].
לכן השאלה הנכונה אינה רק “איך מנקים את הכבד”, אלא מה דוחף אותו אל מחלה, ומה מחזיר אותו אל החלמה. זו אינה רק שאלה של תזונה, אלא של ביולוגיה מערכתית, איכות אנרגטית, דלקת, מנגנוני הגנה ויכולת תיקון [מקור].
הצלחתי להחלים מאי-ספיקת כליות
הגוף אינו זקוק לעוד חומרים.
הוא זקוק לתנאים שבהם הוא יכול להשתמש בהם.
החלמה אינה תוצאה של כוח חיצוני… אלא הענקה של תנאים מדויקים שבהם הגוף חוזר לפעול
פרק 1: הכבד כמראה מערכתית
הכבד הוא אחד האיברים המרכזיים ביותר ברגולציה של חילוף החומרים. הוא מעבד שומנים, סוכרים, חומצות אמינו, תרופות וחומרים רעילים, ולכן הוא מגיב מהר מאוד לשינויים בהרכב התזונה, ברמת הדלקת ובאיזון האנרגטי של הגוף. כאשר יש עומס כרוני, הדבר עלול להופיע תחילה ככבד שומני, ובהמשך כהחמרה תפקודית, דלקתית או פיברוטית [מקור].
חשוב להבין שכבד שומני אינו תמיד “מחלה של הכבד בלבד”. לעיתים הוא הביטוי הגלוי של מערכת שלמה הנמצאת בכיוון לא מאוזן: תנגודת לאינסולין, עומס קלורי, עודף סוכר, שיבוש במיטוכונדריות, הפרעה במיקרוביום, ועקה חמצונית. במילים אחרות, הכבד אינו רק איבר יעד, אלא אינדיקטור של כיוון.
[מקור]
מכאן גם נובעת החשיבות של הסתכלות רחבה: כשמנסים להבין את מצב הכבד, צריך לשאול לא רק מה קורה בכבד עצמו, אלא גם מה קורה במעיים, בשריר, ברקמת השומן, במטבוליזם של הסוכר ובמערכת הדלקתית כולה. ההקשר המערכתי הזה הוא המפתח להבנה עמוקה יותר של מחלת כבד ושל החלמה ממנה [מקור].
פרק 2: צירי המחלה
צירי המחלה בכבד אינם מסתכמים בהצטברות שומן בלבד. ברמה הביולוגית, הם כוללים הפעלה של מסלולים דלקתיים כמו NLRP3 ו-NF-κB, עלייה בעקה חמצונית, פגיעה באיזון המיטוכונדריאלי, שינוי בתקשורת בין תאי הכבד לבין תאי החיסון, והפעלה של תהליכי פיברוזיס [מקור1, מקור2, מקור3, מקור4, מקור5, מקור6].
כאשר NLRP3 מופעל לאורך זמן, הוא תורם להחמרה דלקתית, לגיוס תאי חיסון ולהאצה של תהליכי נזק בכבד. כאשר העקה החמצונית נמשכת, היא פוגעת בממברנות, בחלבונים וב-DNA, ומחלישה את היכולת של התא לתפקד כראוי. כאשר המיטוכונדריות נפגעות, נפגעת גם היכולת של התא לייצר אנרגיה ולבצע תיקון יעיל.
[מקור].
לצד אלה פועלים גם גורמים מטבוליים: עודף סוכר, עודף שומן, תנגודת לאינסולין, נטייה לאגירת שומן בכבד, ולעיתים גם השפעות של אלכוהול ותרופות. אלה אינם רק "גורמי רקע" אלא מנועים פעילים של מחלה. התוצאה המצטברת יכולה להיות מעבר מכבד שומני פשוט, אל דלקת, אל פיברוזיס, ועם הזמן גם אל ירידה עמוקה יותר בתפקוד
פרק 3: צירי ההחלמה
לצד מסלולי המחלה קיימים גם מסלולי החלמה, והם אינם פחות חשובים. Nrf2 הוא אחד ממסלולי ההגנה המרכזיים של התא, ובכבד הוא מסייע להפעיל גנים מגנים, להפחית עקה חמצונית ולחזק עמידות לנזק. מחקרים עדכניים מראים כי הפעלה של Nrf2 קשורה להגנה מפני פגיעה כבדית ולשיפור במצבים של מחלות כבד שונות. [מקור1, מקור2]
גם AMPK ו-SIRT1 הם צירים מרכזיים של החלמה [מקור1, מקור2]. הם קשורים לאיזון אנרגטי, לחילוף חומרים מיטבי, לוויסות דלקת, לשיפור תפקוד מיטוכונדריאלי ולתמיכה במנגנוני תיקון [מקור1, מקור2] ירידה בפעילות של SIRT1 🍇, במיוחד עם הצטברותם של רעלים בגוף, מסיבות שונות, קשורה להחמרה בתפקוד הכבד ולהגברת פגיעות למחלות [מקור]. לעומת זאת, חיזוק הציר הזה עשוי לתמוך בשיקום ובשמירה על יציבות מטבולית [מקור1, מקור2, מקור3, מקור4].
אוטופגיה ומיטופגיה הן חלק מהמערכת הזו. הן מאפשרות לתא לסלק רכיבים פגומים, למחזר חומרים ולשמר איכות מיטוכונדריאלית. בכבד, מנגנונים אלה חשובים במיוחד, משום שהם תורמים לשמירה על הומאוסטזיס, להפחתת נזק מצטבר ולבלימת התקדמות של מחלות מטבוליות ופיברוטיות [מקור1, מקור2, מקור3, מקור4, מקור5].
פרק 4: מה מחזק את ההחלמה בפועל.
יחסי הגומלין ביניהם היא אחת האפשרויות, לדוגמה, הפעלת AMPK (, נושא שעולה כבר בסדרת המאמרים על צירי ההחלמה למשל בחלק ו' ציר החלמה) מעכבת באופן ישיר ומכבה את קומפלקס הדלקת NLRP3 (ציר מחלה). הריפוי מגיח ומכבה את המחלה באופן אוטומטי. חיזוק הריפוי הוא שעומד בפני מחלימני העתיד, גישה זו מבקשת לחשוב מחדש כי היא למעשה מכבה את המחלה אוטומטית. בלימת פיברוזיס מושגת על ידי Nrf2 🥦 ואוטופגיה🧹, המנטרלים תאי כוכב בכבד (Stellate Cells) ומונעים את יצירת הצלקות המבניות [מקור].
כלומר,
כל הרפואה המודרנית, וגם הקלאסית, והרפואה העתיקה כל תרבות ההחלמה המערבית כמעט, מאומנת לחפש:
- מה החומר?
- מה הכדור?
- מה המולקולה?
- מה התרופה?
כלומר: מה הדבר שיזיז את הגוף?
אבל ככל שחוקרים מערכות מורכבות, מתחילה להופיע שאלה אחרת:
מהו המהלך שיאפשר לגוף להזיז את עצמו?
אלה שתי שאלות שונות לחלוטין.
בבואנו למשל להסתכל על המאמר האחרון על הכבד השומני: היום ואז:
לפני עשרים שנים חפשנו, במקרה הטוב, האמת שרק לפני כעשור את:
- הסולפורפאן.
- הרסברטרול.
- Q10.
- חומר שיפעיל NRF2.
והיום, לפחות כפי שאני תופס את זה, מובילי ההחלמה מתחילים לשאול:
- למה NRF2 בכלל כבה?
- למה המיטוכונדריה הפסיקה לעבוד?
- למה DNL הפך למסלול דומיננטי?
- מה השתנה בכיוון התנועה של המערכת?
זאת כבר חשיבה של מהלך.
החלמה אינה נובעת ממסלול אחד בלבד, אלא מסביבה ביולוגית שלמה. תזונה שמפחיתה עומס סוכר ומזונות מעובדים, מפחיתה חלבון מהחי, מפחיתה אלכוהול, ומספקת רכיבי תזונה תומכים, יכולה להוריד את העומס המטבולי מעל הכבד. במקביל, פעילות גופנית סדירה, שינה איכותית וירידה בעקה כללית מחזקות את יכולת הגוף לעבור ממצב של דלקת למצב של תיקון [מקור].

הציור למעלה מציג את SIRT3 כ'מנהל התחזוקה של המנועים'. בזמן שה-AMPK מכבה את האורות כדי לחסוך באנרגיה, ה-SIRT3🍇 זה נכנס פנימה עם מברשת פלדה ומנקה את הפיח מהמנוע (המיטוכונדריה🔋), כדי שכשהאורות יידלקו שוב, והמערכת תעבוד בשיא היעילות [מקור]. צייר ירון מרגולין
לכן, במקום לדבר על “ניקוי כבד” במובן פשטני, נכון יותר לדבר על יצירת תנאים ביולוגיים שמאפשרים לכבד לחזור לתפקוד. זה כולל הפחתת העומס שמפעיל את מסלולי המחלה, וחיזוק התנאים שמפעילים את מסלולי ההחלמה [מקור1, מקור2, מקור3, מקור4, מקור5]
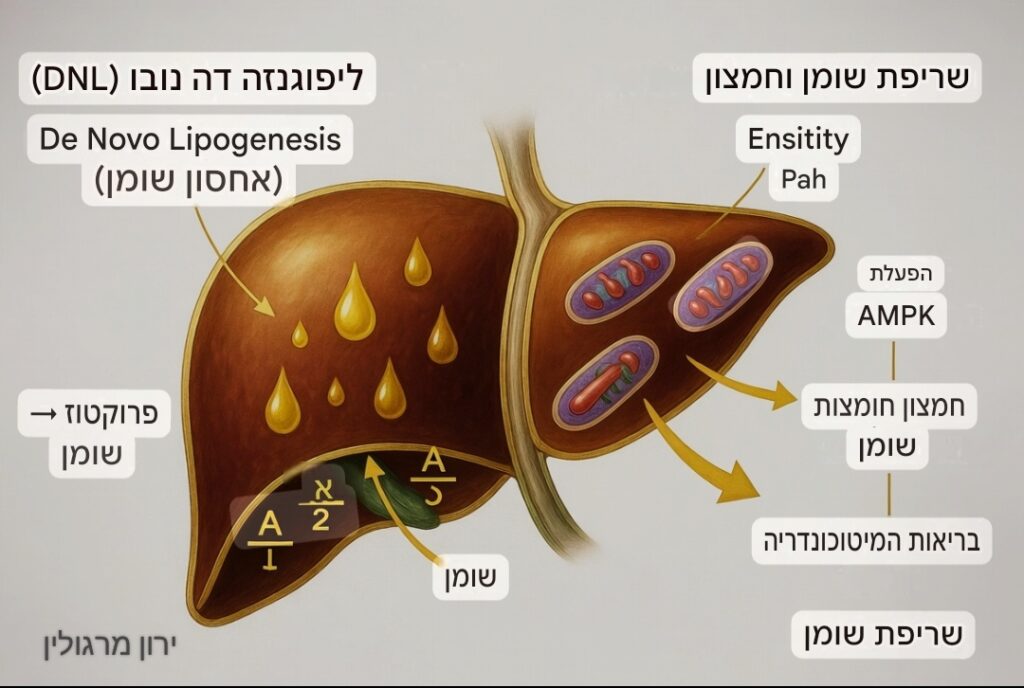
האיור מציג את התהליכים המטבוליים המתרחשים בתוך הכבד, וממחיש את האיזון שבין יצירת שומן לבין פירוקו.
1. האונה השמאלית: ליפוגנזה (יצירת ואחסון שומן) צד זה מדגים כיצד כניסה מוגברת של סוכרים (בדגש על פרוקטוז) מובילה לצבירת שומן בכבד: ליפוגנזה דה נובו (DNL): תהליך שבו הכבד הופך עודפי פחמימות (כמו פרוקטוז) לחומצות שומן חדשות. אחסון שומן: חומצות השומן נאגרות בכבד בצורת טיפות שומן (הטיפות הצהובות באיור), תהליך שעלול להוביל למצב של כבד שומני. מנגנון הבקרה (A2 / A1): הציור מדגים כיצד מסלול מסוים (המסומן בחץ ירוק) מעודד יצירת שומן, בעוד שמסלול מקביל חוסם או מעכב אותו כדי לנסות לשמור על איזון.
2. האונה הימנית: חמצון ושריפת שומן צד זה מציג את התהליך ההפוך – פירוק השומן והפקת אנרגיה: הפעלת AMPK: אנזים המשמש כ"חיישן האנרגיה" של התא. הפעלתו מאותתת לגוף שיש צורך באנרגיה ומעוררת פירוק מאגרי שומן. חמצון חומצות שומן: תהליך שבו חומצות השומן מפורקות כדי לשמש כדלק. בריאות המיטוכונדריה: המיטוכונדריות (האברונים הסגולים באיור) הן "תחנות הכוח" של התא. חמצון השומן מתרחש בתוכן, ותפקודן התקין קריטי למניעת הצטברות שומנים עודפת. שריפת שומן: התוצאה הסופית של המסלול, המפחיתה את עומס השומן בכבד.
פרק 5: שינוי תפיסתי עמוק
המעבר מחשיבה על “ניקוי הכבד” לחשיבה על “צירי מחלה [מקור1, מקור2] וצירי החלמה” הוא לא רק שינוי לשוני. זה שינוי תפיסתי עמוק. הוא מאפשר להבין את הכבד כחלק ממערכת שלמה, לראות את המחלה כתהליך דינמי ולא כגזר דין, ולנסח טיפול שהוא מדויק יותר, רחב יותר ועמוק יותר. [מקור1, מקור2, מקור3]
המשמעות הקלינית והמעשית ברורה: אם אנחנו מבינים איזה מסלולים דוחפים את הכבד לכיוון מחלה, ואיזה מסלולים מחזירים אותו לכיוון של תיקון, אפשר לבנות אסטרטגיה טובה יותר של מניעה, תמיכה והחלמה. הכבד אינו רק איבר שיש “לנקות”; הוא איבר שמספר לנו לאיזה כיוון כל המערכת הולכת [מקור1, מקור2, מקור3].
ההבנה הזו מחזירה את הכבוד למורכבות של הגוף. היא מזכירה שהגוף יודע לתקן, אך הוא זקוק לתנאים הנכונים כדי לעשות זאת. וזה אולי המסר החשוב ביותר: לא רק לטפל במחלה, אלא לאפשר את ההחלמה [מקור].
לסיכום –
אנו מתבוננים אל רפואת העתיד – במקום לדכא סימפטומים, משנים את הסביבה המולקולרית כדי להעיר מסלולים ביולוגיים רדומים. מאפשרים לאדם ליטול אחריות ואפילו לצאת לדרך חופשית עם מצפון וכיוון לעבודה מעשית.
🔹 מסלולי ההחלמה -המסלולים הביולוגיים שמשקמים את הגוף – חלק א' מאת ירון מרגולין
נשארו לך שאלות
🔬אשמח להשיב על כל שאלה
בבקשה לא להתקשר משום שזה פשוט לא מאפשר לי לעבוד – אנא השתמשו באמצעים שלפניכם
למען הסר ספק, חובת התייעצות עם רופא (המכיר לפרטים את מצבו הבריאותי הכללי של כל מטופל או שלך) לפני שימוש בכל תכשיר, מאכל, תמצית או ביצוע כל תרגיל. ירון מרגולין הוא רקדן ומבית המחול שלו בירושלים פרצה התורה כאשר נחשפה שיטת המחול שלו כבעלת יכולת מדהימה, באמצע שנות ה – 80 לרפא סרטן. המידע באתר של ירון מרגולין או באתר "לחיצות ההחלמה" (בפיסבוק או MARGOLINMETHOD.COM ), במאמר הנ"ל ובמאמרים של ירון מרגולין הם חומר למחשבה – פילוסופיה לא המלצה ולא הנחייה לציבור להשתמש או לחדול מלהשתמש בתרופות – אין במידע באתר זה או בכל אחד מהמאמרים תחליף להיוועצות עם מומחה מוכר המכיר לפרטים את מצבו הבריאותי הכללי שלך ושל משפחתך. מומלץ תמיד להתייעץ עם רופא מוסמך או רוקח בכל הנוגע בכאב, הרגשה רעה או למטרות ואופן השימוש, במזונות, משחות, תמציות ואפילו בתרגילים, או בתכשירים אחרים שנזכרים כאן.
physician (who knows in detail the general health of each patient or yours) before using any medicine, food, extract or any exercise. The information on Yaron Margolin's website or the "Healing Presses" website (on Facebook or YARONMARGOLIN.COM), in the above article and in Yaron Margolin's articles are material for thought – philosophy neither recommendation nor public guidance to use or cease to use drugs – no information on this site or anyone You should always consult with a qualified physician or pharmacist regarding pain, bad feeling, or goals and how to use foods, ointments, extracts and even exercises, or other remedies that are mentioned as such
מאמרים אחרונים
- הכבד כמראה מערכתית: צירי מחלה מול צירי החלמה
- חלק ו' על צירי ההחלמה ומה שמפעיל אותם – (signal architecture) מאת ירון מרגולין
- 🔹 מסלולי ההחלמה -המסלולים הביולוגיים שמשקמים את הגוף – חלק א' מאת ירון מרגולין
- המסלולים הביולוגיים שמשקמים🔹 את הגוף – חלק ב', על ההחלמה מאת ירון מרגולין
- המסלולים הביולוגיים שמשקמים את הגוף – חלק ג', על ההחלמה
- 🔷 המסלולים הביולוגיים שמשקמים את הגוף: p53 — שומר הגנום – חלק ד’ — ירון מרגולין
- חלק ה' – חלבוני ההחלמה: הרשת המולקולרית של הריפוי, ההתחדשות וההומאוסטזיס מאת ירון מרגולין
- המסלולים הביולוגיים המשקמים את הגוף: פרספקטיבה חדשה על החלמה
- 📘 The Biological Pathways That Restore the Body: A New Perspective on Recovery
- גמישות העורקים – רפואת העתיד – חלק א'
- עורקים גמישים חלק ב', הסבר מפורט על הצירים. הביולוגיה העמוקה של גמישות העורקים מאת ירון מרגולין
- קלוטו – האם קלוטו הוא מעיין הנעורים הזורם במערה מוסתרת וסודית?
- המחמצן הגדול – Ros ודרכי ההתגוננות מפניו ללא תרופות
- חימצון האינסולין והגברה של חומצת שתן בדם, גאוט, אי ספיקת כליות, שבץ לב וסוכרת
- מיטוכונדריה – במיטוכונדריון מתחילה מחלת הכליות
- ירידה בתפקוד הכליות – מה לעשות?
- דיוקסינים במזון
- כשהמוסר מתנתק מן החיים: כיצד הפכה האידאולוגיה הנאורה מביקורת מוסרית לעמדה הרסנית ומשתקת
- קדרה ואבדון
- Cauldron and Ruin
- אוכלוסיית המעי הגס חיידקים טובים, דיסביוזיס ו “שומר הגנום” גן p53
- מחסור בברזל
- מיומטי תנועה – התקווה החדשה לחולי כליות: תרכובות שמחקות את צירי ההחלמה של פעילות גופנית
- בשר, כליות וזרימת דם מואצת: כיצד תזונה מהחי פוגעת במערכת הסינון של הגוף
- אמנות ההחלמה האבודה
- קרקר מדהים מכוסמת ירוקה – מתכון
- השפעה קלינית של ריבס על הטיפול באי ספיקת כליות כרונית: כפי שמשתקף במחקרים.
- גמישות העורקים – רפואת העתיד – חלק א'
- עורקים גמישים חלק ב', הסבר מפורט על הצירים. הביולוגיה העמוקה של גמישות העורקים מאת ירון מרגולין
- קלוטו – האם קלוטו הוא מעיין הנעורים הזורם במערה מוסתרת וסודית?
- דיוקסינים במזון
- קדרה ואבדון
- Cauldron and Ruin
- אוכלוסיית המעי הגס חיידקים טובים, דיסביוזיס ו “שומר הגנום” גן p53
- מחסור בברזל
- מיומטי תנועה – התקווה החדשה לחולי כליות: תרכובות שמחקות את צירי ההחלמה של פעילות גופנית
- בשר, כליות וזרימת דם מואצת: כיצד תזונה מהחי פוגעת במערכת הסינון של הגוף
- קרקר מדהים מכוסמת ירוקה – מתכון
- השפעה קלינית של ריבס על הטיפול באי ספיקת כליות כרונית: כפי שמשתקף במחקרים.
- טסטוסטרון אינו גורם, מזרז או מחמיר סרטן ערמונית – וה-FDA מתחיל להודות בזה
- הקשר בין כליה לבריחת סידן – תפקוד לא מוערך של הכליה במניעת אוסטאופורוזיס
- על הגאונות והגאון
- 📘 The Biological Pathways That Restore the Body: A New Perspective on Recovery
- הגוף יודע על המחלה: כיצד לזהות אותה באמצעות מבט עומק בבדיקות המעבדה ובתסמינים הגופניים
- טאורין: חומצה אמינית מסתורית ומסקרנת
- מלטונין הוכח שהורמון השינה נוגד חמצון רב עוצמה ויעיל כתרופה בקרב חולי כליה
- מיטוכונדריה – במיטוכונדריון מתחילה מחלת הכליות
- סמנים ביולוגיים (Novel Biomarkers) ובדיקת כליות מוק
- איך התפתחו לחיצות ההחלמה בבית מחול ירון מרגולין בירושלים
- המזון הבסיסי והחומצי – מה כן ומה לא נכון. מה עובד על ההחלמה ומה הן אגדות? חומציות מגיעה מתזונה והיא קמה והורסת את הכליות
- הביוכימיה של ההחלמה: מפת העל של החיים
- על הגאונות והגאון
- 📘 The Biological Pathways That Restore the Body: A New Perspective on Recovery
- הגוף יודע על המחלה: כיצד לזהות אותה באמצעות מבט עומק בבדיקות המעבדה ובתסמינים הגופניים
- איך התפתחו לחיצות ההחלמה בבית מחול ירון מרגולין בירושלים
- המזון הבסיסי והחומצי – מה כן ומה לא נכון. מה עובד על ההחלמה ומה הן אגדות? חומציות מגיעה מתזונה והיא קמה והורסת את הכליות
- הביוכימיה של ההחלמה: מפת העל של החיים
- הבדידות – חזית חדשה ומקור רב משמעות למחלות כרוניות – מגפת הבדידות
- השמנה בטנית – סטרס ביולוגי — מנגנון העל שמסביר השמנה בטנית גם בלי אכילת יתר. למה לא יורדים בבטן בתהליכי הרזיה מסודרים?
- שבריריות גופנית מחדל רפואי – או אובדן רזרבה
- פאסיה – חיתולית Fascia, שיטות חדשות להפחתת כאב
- אנמיה – רוצה לחשוב מחדש על אנמיה ללא סיכון מתוספי ברזל מחמצנים?
- מחלה כרונית קשה עכשיו – סבלנות
- דיאליזה: מאבק הירואי שאינו ריפוי
- השפעה קלינית של ריבס על הטיפול באי ספיקת כליות כרונית: כפי שמשתקף במחקרים.
- טסטוסטרון אינו גורם, מזרז או מחמיר סרטן ערמונית – וה-FDA מתחיל להודות בזה
- הקשר בין כליה לבריחת סידן – תפקוד לא מוערך של הכליה במניעת אוסטאופורוזיס. גלו את תפקיד קולטן VDR בכליות
- כשהמוסר מתנתק מן החיים: כיצד הפכה האידאולוגיה הנאורה מביקורת מוסרית לעמדה הרסנית ומשתקת
- מכתב תודה ממחלימת כליות בתוך ארבעה חודשים.
- השמנה בטנית – סטרס ביולוגי — מנגנון העל שמסביר השמנה בטנית גם בלי אכילת יתר. למה לא יורדים בבטן בתהליכי הרזיה מסודרים?
- שבריריות גופנית מחדל רפואי – או אובדן רזרבה
- פאסיה – חיתולית Fascia, שיטות חדשות להפחתת כאב
- מחלה כרונית קשה עכשיו – היו סבלניים.
- הקשר בין כליה לבריחת סידן – תפקוד לא מוערך של הכליה במניעת אוסטאופורוזיס
- מקצבים בעולם החי – שעון ביולוגי בתאי הגוף
- מזון למוח – המזון הבריא למוח תומך בגמישות מערכת העצבים שלו וביכולת הלמידה, שומר על הזכרון, ומונע מחלות כגון אלצהיימר.
- מרחבי חיים מאריכי חיים – האזורי הכחולים.
- עשרת המזונות הבריאים ביותר לחולי כלייה על האצות והפוקוקסנטין, חלק 7.
- אילוף הכליות הסוררות
- סגולותיהם של זרעי החרדל
- דרכים חדשות לוויסות יעיל של טריגליצרידים (Triacylglycerols) בדם
- שוויון בריאותי?
- האם תזונה נוגדת דלקת יכולה לעזור?
- חידושים ומהפכות בנושא יתר לחץ דם.
- נוגדני ANF – גורם אנטי גרעיני ונוגדן אנטי-ציטופלזמי של נויטרופיל – ANCA
- תעוקת חזה מיקרו-וסקולרית
- "מגיפת המאה העשרים ואחת", מיתוסים רפואיים: כל מה שחשוב לדעת על אלרגיות
- מזון כתרופה
- הלשון של הכליות
- כיצד הכליה מטפלת בחלבונים
- אינסולין ותפקיד הכליות במטבוליזם ופינוי
- עוצמתם של נבטי הברוקולי כתרופה לחולי כליה
- תפקוד הכליה כבלוטה הורמונלית: סקירה מדעית
- כיצד הכליה מטפלת בחלבונים
- אנטיביוטיקה – על נזקיה לאנשים בכלל ובפרט לחולים
- תזונה תאית
- כל מה שאתה צריך לדעת על מיקרו-תזונה – ויטמינים ומינרלים
- להתסיס שיבולת שועל – מתכון וכל הסודות.
- כל מה שאתה צריך לדעת על מיקרו-תזונה – ויטמינים ומינרלים
- להתסיס שיבולת שועל – מתכון וכל הסודות
- למה מדידת הויטמין B12, רחוקה מלהיות מדויקת?
- הומוציסטאין, ויטמינים ומניעת מחלות כלי דם
- ירקות ירוקי עלים לרוב טובים לבריאותנו, לפעמים הם לא – רוצה לדעת מתי כדאי לצרוך עלים ירוקים?
- תרופות ללחץ דם גבוהה נמצאו כקשורות לסיכון מוגבר לסכיזופרניה
- חומץ אננס – רוצה לדעת כיצד להכין תרופה ביתית, אנטי-דלקתי מקליפות האננס?
- תחמוצת החנקן (NO) כיווץ תאי האנדותל, מחלות כרוניות קשות – ודרכי ריפוי חדשות
- תרופה ישראלית חדשה לקורונה
- יתר לחץ דם – שינוי פרדיגמה וגישות חדשות – לחץ דם גבוה וטיפול טבעי ללא תרופות לאחר שטיפול תרופתי נערץ ככל הנראה כשל
- מזון כתרופה – שמן זית ושמן קנולה או חרדל הצילו מאות ילדים בעולם.
- מחשבון מדידה והערכת קצב הסינון הגלומרולרי (GFR) אשר מבוסס על קריאטינין, נתוני גוף, גיל, גזע ומין.
- מזון כתרופה – הצלחתי להחלים מאי ספיקת כליות
- עשרת המזונות הבריאים ביותר לחולי כלייה – על השעורה (דגן) ופעולות נוגדות דלקת כולל עיכוב גורם נמק גידול אלפא – Tumor necrosis factor alpha (TNF-α).
- על הנקמה ודמימותיה
- גודש נוזלים בריאות – בקרב חולי כליה
- איך להיראות צעיר מגילך – כל הסודות
- על ההשפעה האיומה של חלבון מן החי על הכליות
- עשרת המזונות הבריאים ביותר לחולי כלייה – קמח קליפת רימונים (PPF) .9
- Reconstruction of erythropoietin by the kidneys without drugs
- קטואנאלוג ketoanalogue
- תרופות ללחץ דם גבוהה נמצאו כקשורות לסיכון מוגבר לסכיזופרניה
- חומץ אננס – רוצה לדעת כיצד להכין תרופה ביתית, אנטי-דלקתי מקליפות האננס?
- גלוטמין (Gln) -המגן הגדול על בריאות האדם – כל מה שחשוב לדעת
- פיקנוגנול – כל האמת על רפואת עץ האורן והכנת התה ממחטיו
- גלוטתיון כל האמת
- הגיל השלישי, תאוותיה של הזיקנה – ואיך להתבגר יפה, טוב ובריא
- הבדידות – חזית חדשה ומקור רב משמעות למחלות כרוניות – מגפת הבדידות
- התערבויות בניהול רגשות, מחשבות ואורח חיים כדי להתמודד עם מגיפה של מחלות כליתיות
- חומוס מזין, מרגיע ונוגד חרדות – מתכונים
- בני-על – האם יש גבול ביולוגי למספר השנים שאדם יכול לחיות?
- 8 חסרים תזונתיים שכיחים
- האם בוטנים, קליפות בוטנים ומוצריהן הם מזון על?
- להתסיס שיבולת שועל – מתכון וכל הסודות
- סיבי תזונה
- תנגודת לאינסולין וקמח מפרי עץ החרוב
- קמח קליפות בצל ותה קליפות בצל – מתכון
- אבחון מוקדם למחלת הכליות יכול להציל חיי אדם
- פורינים חוץ תאיים ותחזוקת הכליות
- פחד גבהים – מהו פחד גבהים וטיפול
- חומצת שתן נמוכה, או היפואוריצמיה (Hypouricemia)
- התערבויות בניהול רגשות, מחשבות ואורח חיים כדי להתמודד עם מגיפה של מחלות כליתיות
- פחות מתים – טכניקת דיאליזה חדשה שבולמת את קצב המתים ומגבירה את ההישרדות בכשל כליות.
- נאטו (פולי סויה מותססים יפניים) מתכון
- מיטוכונדריה – במיטוכונדריון מתחילה מחלת הכליות
- התקף לב / אירוע לבבי בעקבות מחלת כליות – מסלול איתות שמגן על הלב בשם "VEGFA" משתבש בגלל מחלת כליות כרונית.
- לראשונה, מדענים הצליחו לתרגם לטקסט את הפעילות המוחית של אדם עם שיתוק שאינו מסוגל לדבר.
- טיפולים מבוססי רירית לזיהומים נגיפיים וחיידקיים
- נוגדני ANF – גורם אנטי גרעיני ונוגדן אנטי-ציטופלזמי של נויטרופיל – ANCA
- תעוקת חזה מיקרו-וסקולרית
- "מגיפת המאה העשרים ואחת", מיתוסים רפואיים: כל מה שחשוב לדעת על אלרגיות
- מזון כתרופה
- חומציותו של הנוזל הבין תאי בגוף האדם מונעת תהליכי החלמה במחלות כרוניות קשות – מה לעשות?
- עריכת גנים מותאמת אישית, in vivo, עבור אדם חולה שהצליחה – מהפכה בעולם הרפואה
- אנטיביוטיקה – על נזקיה לאנשים בכלל ובפרט לחולים
- תזונה תאית
- כל מה שאתה צריך לדעת על מיקרו-תזונה – ויטמינים ומינרלים
- להתסיס שיבולת שועל – מתכון וכל הסודות.
- סוויטה לירית" (Lyric Suite) אנא סוקולוב